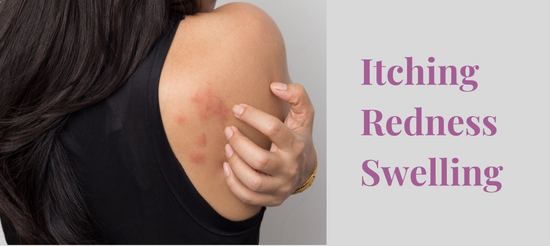
Skin Care Ingredients That Cause Allergic Reactions on the Face
There are many skin care ingredients that cause allergic reactions on the face. The only way to prevent this from happening again is to figure out which ingredients caused the rash. Then, you can read product labels to avoid that ingredient.
If you have had a face allergy to a skin cream or new skincare product, you know how uncomfortable the skin inflammation from a skin allergy can be. Whether you have tiny bumps on the face, redness, swelling, hives or itching, the treatment is the same. You need to soothe and calm the skin and avoid the allergenic ingredient.
You can treat the face allergy with anti-inflammatory and soothing ingredients.
However, you must figure out what skin care ingredients in your skin care products that caused the skin allergy.

Cosmetic Ingredients That Cause Allergies
If you have an allergic reaction on your face, chances are it is one of these 6 types of ingredients that are causing it. A rash on the eyelids is almost always due to nail polish, while a rash on the hands is usually hand soap. An allergic reaction on the face can be caused by many ingredients, but fragrances and preservatives are the most common.
The 6 most common ingredients in skin care products that cause allergic reactions on the face are:
- Fragrances
- Essential Oils
- Preservatives
- Plant derived ingredients
- Chemical sunscreens
- Parabens
How To Figure Out What Skin Care Ingredients I Am Allergic To?
If you suffer from a skin allergy from a skin care product you have three choices:
- Figure out what you are allergic to by keeping a diary
- See a dermatologist and get patch testing
- Find a skincare brand that is hypoallergenic
If you get an allergic reaction, start a diary and write down every ingredient on the ingredient list in any skin care products that you used over the last 48 hours. Then match the list to these common allergen lists. Below you can find lists of the most common allergens in skin care products.
Circle the items in your diary that are on the allergenic ingredient list.
With time you will notice a pattern and will see that it is always the same ingredient that is giving you a rash.
You can also find a dermatologist at aad.org and get a patch test (Not all dermatologists do patch testing so ask before you schedule an appointment).
Hypoallergenic Skin Care Brands
Our favorite hypoallergenic skincare brands are:
- VMV Hypoallergenics- was developed by a dermatologist and does not have the 100 most common allergens in it.
- Vanicream
- La Roche Posay Toleriane products

Allergy to Niacinamide
Niacinamide can cause a skin allergy. We have see happen with EltaMD UV Clear SPF and other niacinamide containing products.
Rag Weed Allergy
If you have a ragweed allergy, certain skincare ingredients may cause cross-reactions due to their botanical relations or similarity in allergenic components. It's essential to be cautious and potentially avoid ingredients that are in the same family as rag weed.
Skin care ingredients to avoid if you have a ragweed allergy:
- Chamomile – This popular soothing ingredient is related to ragweed.
- Echinacea – Often used for its anti-inflammatory properties.
- Marigold (Calendula) – Used for its soothing and healing properties.
- Sunflower oil – Sometimes found in moisturizers and other skincare products.
- Helenium – Used in some herbal skincare products.
- Dandelion extract – Occasionally used in natural or botanical products.
- Tansy oil – Found in some herbal formulas.
- Yarrow – Used for its astringent properties.
- Artemisia – Another name for this is wormwood or sagebrush, and it's sometimes used in skincare.

Allergy to Preservatives
If you have a preservative allergy, certain preservatives in skincare and cosmetic products can cause allergic reactions.
Skin care ingredients to avoid if you have a preservative allergy:
- Parabens (e.g., methylparaben, ethylparaben, propylparaben, butylparaben)
-
Formaldehyde and formaldehyde-releasing preservatives:
- Quaternium-15
- DMDM hydantoin
- Imidazolidinyl urea
- Diazolidinyl urea
- Polyoxymethylene urea
- Sodium hydroxymethylglycinate
- Methylisothiazolinone (MIT) and Methylchloroisothiazolinone (CMIT)
- Benzyl alcohol
- Phenoxyethanol
- Bronopol (2-bromo-2-nitropropane-1,3-diol)
- Isothiazolinones
- Thimerosal
- Triclosan and Triclocarban
- Propylene glycol and butylene glycol (though these are more commonly classified as irritants, they can sometimes cause allergic reactions)
- Methyldibromo glutaronitrile/phenoxyethanol (Euxyl K 400)

Fragrance Allergy
If you have a fragrance allergy, you should be aware that fragrances are among the top five allergens in cosmetics and other products. People sensitive to fragrances should look out for a wide array of ingredients, as the term "fragrance" or "parfum" in an ingredient list can actually be a blend of many components.
What skin care ingredients to avoid if you have a fragrance allergy:
- Fragrance or Parfum: A catch-all term that can refer to a blend of multiple fragrance ingredients.
- Linalool
- Limonene
- Eugenol
- Cinnamal (Cinnamaldehyde)
- Geraniol
- Isoeugenol
- Coumarin
- Citral
- Benzyl Alcohol (not always used as a fragrance but can be present in some fragrant blends)
- Benzyl Salicylate
- Benzyl Benzoate
- Farnesol
- Citronellol
- Hexyl Cinnamal
- Amyl Cinnamal
- Cinnamyl Alcohol
- Oakmoss and Treemoss Extracts (Evernia prunastri and Evernia furfuracea)
- Balsam of Peru (or Myroxylon pereirae) – it's both a fragrance and flavoring agent, and can be found in many products.
- Essential oils – even though they're natural, they can still cause reactions in sensitive individuals. Lavender, peppermint, tea tree, and ylang-ylang are common essential oils in skincare products.
Additionally, "unscented" products might still contain fragrances to mask the scent of other ingredients. If you're sensitive, opt for products labeled "fragrance-free."
Remember, sensitivity to one fragrance ingredient doesn't mean you'll react to them all. But given the complexity and lack of transparency in fragrance formulations (they're considered trade secrets), many people with fragrance allergies opt to avoid all fragranced products.

Hydroquinone Allergy
Hydroquinone allergy is not uncommon. You may get a rash after using a prescription skin lightening product. If you are allergic to hydroquinone, you need to avoid all hydroquinone derivatives.
You may also be allergic to these hydroquinone-derived cosmeceuticals:
- arbutin
- kojic acid

These are the most common skincare ingredients that cause a skin allergy (in alphabetical order):
2,5 Diaminotoluene Sulfate
2,6-Ditert-butyl-4-cresol (BHT)
2-Ethylhexyl-4-dimethylaminobenzoate (Eusolex 6007) (Padimate O) (Octyl Dimethyl paba)
2-Ethylhexyl-4-methoxycinnamate (Parsol MCX)
2-Hydroxy-4-methoxy-4-methylbenzophenone (Mexenone)
2-Hydroxy-4-methoxy-benzophenon-5-sulfonic acid (Sulisobenzone)
2-Hydroxy-4-methoxybenzophenone (Eusolex 4360)
2-Nitro-P-Phenylenediamine
2-tert-Butyl-4-methoxyphenol (BHA)
3-(4-Methylbenzyliden)camphor (Eusolex 6300)
3-Aminophenol Hydroquinone
4-Aminobenzoic acid (PABA)
4-Aminophenol
4-Chloro-3-cresol (PCMC)
4-tert-Butyl-4’-methoxydibenzoylmethane (Parsol 1789) (Avobenzone)
Aloe vera
Ammonium Persulfate
Ammonium Thioglycolate
Angelica
Arnica
Balsam of Peru (myroxylon pereirae)
Beeswax
Benzoic Acid
Benzophenone-3 (oxybenzone)
Benzyl alcohol
Benzyl salicylate
Bladderwrack
Calendula (Marigold)
Camphor
Carmine
Catnip
Centella (gotu-kola)
Cetyl alcohol
Chamomile
Chloracetamide
Chlorhexidine gluconate
Cocamide DEA
Cocamidopropyl Betaine
Colophony (rosin)
Compositae Mix
Coriander
Cucumber
Dog rose hips
Echinacea
Euxyl K 400 (Methyldibromo-glutaronitrile-phenoxyethanol)
Formaldehyde
Formaldehyde Releasing Preservatives (FRPs):
Quaternium 15
Imidazolidinyl Urea (Germall)
Diazolidinyl Urea (Germall II)
Bromonitropropane diol (Bronopol)
DMDM Hydantoin
Gingko
Glyceryl Thioglycolate
Glyceryl Thioglycolate
Goldenseal
Homomenthylsalicylate (Homosalate)
Hops
Hydrogen Peroxide
Hydroxydecyl Ubiquinone (Idebenone)
Isoamyl-p-methoxycinnamate
Isopropyl myristate
Kelp
Lanolin alcohol
Lavender
Marigold
Methoxycinnamate
Methyl Methacrylate
Methylchloroisothiazolone (MCI)
Methylisothiazolinone
Niacinamide- some unpure forms cause an allergic reaction while pure forms do not.
Octyl salicylate (Octisalate)
Parabens
Para-phenylenediamine (PPD)+
Phenylbenzimidazol-5-sulfonic acid (Eusolex 232)
Potassium Dichromate
Propolis (bee’s glue)
Propyl gallate
Propylene Glycol
P-Tert-Butylphenol Formaldehyde Resin
Rosemary (Rosmarinus Officinalis (Rosemary) Leaf Extract)
Sage
Sesquiterpene Lactone
Sodium Benzoate
Sorbic Acid
Sorbitan monooleate (Span 80)
Sorbitan sesquioleate
St. John’s wort
Stearyl alcohol
tert-Butylhydroquinone
Toluenesulphonamide Formaldehyde Resin (tosylamide)
Triclosan (Irgasan DP 300)
Triethanolamine
Witch hazel
Ylang Ylang Oil
Natural Plant Ingredients That Can Cause Skin Allergy
Natural and organic products are common causes of skin allergy. Here are some plant derived skin care ingredients that commonly cause skin allergy:
Aloe vera
Angelica
Arnica
Balsam of Peru (Myroxylon pereirae)
Beeswax
Bladderwrack
Catnip
Colophony (rosin)
Compositae Mix
Coriander
Cucumber
Dog rose hips
Echinacea
Ginkgo
Goldenseal
Gotu kola (Centella asiatica)
Hops
Kelp
Lavender
Licorice
Marigold
Propolis (bee’s glue)
Rosemary
Sage
Sesquiterpene lactone
St. John’s wort
Witch hazel
Ylang-ylang oil
Sunscreen Ingredients That Cause Skin Allergy
2-Ethylhexyl-4-dimethylaminobenzoate (Eusolex 6007) (Padimate O) (Octyl Dimethyl paba)
2-Ethylhexyl-4-methoxycinnamate (Parsol MCX)
2-Hydroxy-4-methoxy-4-methylbenzophenone (Mexenone)
2-Hydroxy-4-methoxy-benzophenon-5-sulfonic acid (Sulisobenzone)
2-Hydroxy-4-methoxybenzophenone (Eusolex 4360)
3-(4-Methylbenzyliden)camphor (Eusolex 6300)
4-Aminobenzoic acid (PABA)
4-tert-Butyl-4’-methoxydibenzoylmethane (Parsol 1789) (Avobenzone)
Benzophenone-3 (oxybenzone)
Homomenthylsalicylate (Homosalate)
Isoamyl-p-methoxycinnamate
Octyl salicylate (Octisalate)
Phenylbenzimidazol-5-sulfonic acid (Eusolex 232)
Preservatives that cause skin allergy
Benzoic acid
Benzyl alcohol
Euxyl K 400 (Methyldibromo glutaronitrile and phenoxyethanol)
Formaldehyde-releasing-preservatives
Quaternium 15
Imidazolidinyl urea (Germall)
Diazolidinyl urea (Germall II)
Bromonitropropane diol (Bronopol)
DMDM hydantoin
Methylchloroisothiazolinone and Methylisothiozolinone (MCI/MI)
P-tert-Butylphenol formaldehyde resin
Parabens
Propylene glycol
Sodium benzoate
Toluenesulphonamide Formaldehyde
Resin (tosylamide)
Fragrance in Skin Care That Causes Skin Allergy
Balsam of Peru
Fragrance Mix 1
Fragrance Mix 2
Cinnamic alcohol
Eugenol
Cinnamic aldehyde
Isoeugenol
Geraniol
Lyral
Ylang-ylang
Hydroxycitronella
Oak moss absolute
Benzyl Alcohol
Narcissus
Jasmine
Citral
Sandalwood
Farnesol
Citronellol
Tea tree
a-Hexyl-cinnamic aldehyde
Coumarin
α-amyl cinnamic aldehyde
Our advice is to write down everything you put on your face in the last 12 hours. With time you will start to see a pattern and this can help you narrow down which ingredients caused the reaction. You can also see a dermatologist and get patch testing done to identify the exact allergens.
It is so difficult to figure out what you are allergic to. You may even be allergic to more than one skin care ingredient. So if this is a concern for you- get patch testing done by your dermatologist. It will make buying skin care so much easier for you!

What ingredient in lipstick causes an allergic reaction?
Carmine is a reddish color made from crushed insects. Many people are allergic to it. It is found in lipsticks, blushes and other red makeups.
What ingredient in mascara causes an allergy?
Formaldehyde in mascara commonly causes a mascara allergy.
What ingredient in nail polish causes an allergy?
Formaldehyde and toluene in nail polish can cause an allergy. The rash from nail polish allergy usually causes redness and scaling of the eyelids. This is from touching the eyelids when you are wearing nail polish that you are allergic to.
What is the allergen in hand wash?
Triclosan is an antimicrobial ingredient in hand soaps that often causes hand dermatitis.